This makes GIS more suitable for certain purposes such as indoor placement, as opposed to air-insulated electrical gear, which takes up considerably more room. This property makes it possible to significantly reduce the size of electrical gear.

The high dielectric strength is a result of the gas's high electronegativity and density.

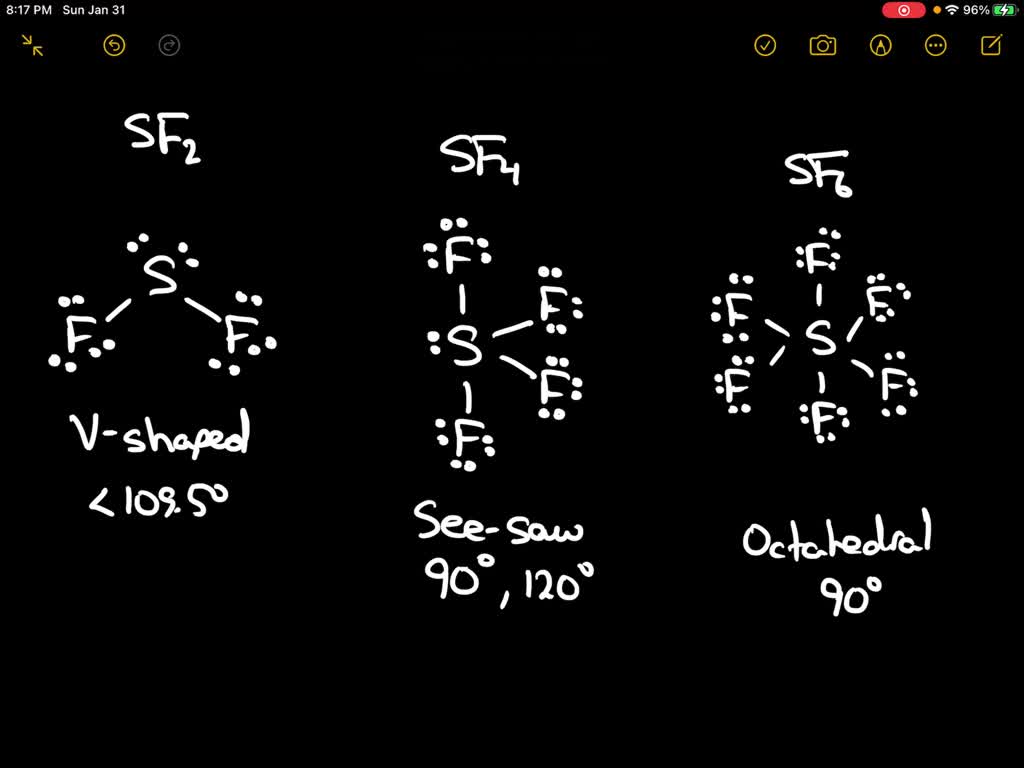
SFĦ gas under pressure is used as an insulator in gas insulated switchgear (GIS) because it has a much higher dielectric strength than air or dry nitrogen. Dielectric medium Ħ is used in the electrical industry as a gaseous dielectric medium for high-voltage sulfur hexafluoride circuit breakers, switchgear, and other electrical equipment, often replacing oil-filled circuit breakers (OCBs) that can contain harmful polychlorinated biphenyls (PCBs). Other main uses as of year 2015 included a silicon etchant for semiconductor manufacturing, and an inert gas for the casting of magnesium. The electrical power industry used about 80% of the sulfur hexafluoride produced in year 2000, mostly as a gaseous dielectric medium. It does not react with molten sodium below its boiling point, but reacts exothermically with lithium. A main contribution to the inertness of SF 6 is the steric hindrance of the sulfur atom, whereas its heavier group 16 counterparts, such as SeF 6 are more reactive than SF 6 as a result of less steric hindrance (See hydrolysis example). There is virtually no reaction chemistry for SFĦ. Uses in magnesium, aluminium, and electronics manufacturing also hastened atmospheric growth. The increase over the prior 40 years was driven in large part by the expanding electric power sector, including fugitive emissions from banks of SFĦ gas contained in its medium- and high-voltage switchgear. It is generally transported as a liquefied compressed gas.Ħ in Earth's troposphere reached 10.66 parts per trillion (ppt) in year 2021, rising at 0.36 ppt/year. It has a density of 6.12 g/L at sea level conditions, considerably higher than the density of air (1.225 g/L). It is a hypervalent molecule.Ħ is poorly soluble in water but quite soluble in nonpolar organic solvents. SFĦ has an octahedral geometry, consisting of six fluorine atoms attached to a central sulfur atom. It is inorganic, colorless, odorless, non- flammable, and non-toxic. This is as opposed to water where the partial positive hydrogen poles are more attracted to the negative lone pair electron region.Sulfur hexafluoride (SF 6) or sulphur hexafluoride ( British spelling) is an extremely potent and persistent greenhouse gas that is primarily utilized as an electrical insulator and arc suppressant. This is most likely also a result of the more negative fluorine dipoles which themselves are repelled from the lone pair electrons by negative-negative charge repulsion. The bonding angle for SF2 is approximately 98˚ which is less than that of water (H2O) which has a bonding angle of 104.5˚. What else is interesting about this compound?
SF2 COMPOUND FREE
Feel free to check out the Lewis Dot Structure of H2O and information related to its polarity. the newly formed molecule has many more electrons) and because the region of negative charge due to lone pairs has been essentially removed/cancelled away by the double bond.įor a reference point, the structure of SF2 is similar to that of H2O in terms of its configuration due to the same kind of electron-electron repulsion. However, these numbers are probably not representative of those for SF2 because of the much larger structure (i.e. S2F4 has a melting point of -98˚C and a boiling point of 39˚C. In this new structure two SF2 molecules combine and form a double bond in between them. Due to the opposing regions of negative charge (one due to electronegativity and another due to lone pair electrons), the molecule is extremely unstable and readily "decomposes" into the molecule S2F4. Although this author attempted to locate verifiable information about the boiling and freezing point of this molecule, this was not readily available due to the inherent instability present within the molecule. Answer: SF2 is a polar molecule due to the presence of lone pair electrons on sulfur which force the molecule to adopt a bent configuration due to electron-electron propulsion.ĭue to the difference in electronegativity between Fluorine (3.98) and Sulfur (2.58), the molecule has a weaker dipole moment because some negative charge is being pulled towards the fluorine ends causing a little bit more of an even distribution of charge within the molecule.
